
- Home
- Brand
- Connectivity
- Model
- Bcd325p2 (52)
- Bcd396xt (28)
- Bcd436hp (291)
- Bearcat (19)
- Cms600p2 (500)
- Cms600p2 Vet (23)
- Cms600p2-vet (79)
- Cms600p2vet (88)
- Ds3608-sr00003vzww (19)
- Handscan V8 (26)
- High Resolution (121)
- Pro-106 (39)
- Pro-651 (32)
- Pro-668 (41)
- Pro-96 (32)
- S50a (24)
- Sds100 (155)
- Trx-1 (128)
- V7 For Veterinary (21)
- Ws1040 (80)
- ... (2280)
- Scan Engine
- Series
- 1470g (2)
- Ds3508 (7)
- Ds3508-er (3)
- Ds3578 (2)
- Ds3608 (8)
- Ds3608-dp (2)
- Ds3608-sr (14)
- Ds4308 (5)
- Ds4308-hd (3)
- Honeywell Xenon 1950 (2)
- Li3608-er (3)
- Memor X3 (6)
- Motorola Ds3508 (3)
- Philips Dpm (3)
- Symbol Ds6707 (7)
- Symbol Ls2208 (2)
- Voyager 1472g (4)
- Zebra Ds3608 (5)
- Zebra Ds3608-sr (11)
- Zebra Li3608 (6)
- ... (3980)
- Type
- Bar Code Scanner (6)
- Barcode Scanner (118)
- Barcode Scanners (12)
- Calling System (30)
- Earpiece (9)
- Hand Held (22)
- Handheld (169)
- Handheld Scanner (44)
- Mobile (10)
- Mobile / In-vehicle (6)
- Mobile Scanner (8)
- Parts & Accessories (9)
- Portable (19)
- Portable / Handheld (662)
- Radio Scanner (8)
- Radio Scanners (9)
- Scanner (64)
- Ultrasound Machine (6)
- Ultrasound Scanner (10)
- White And Black (6)
- ... (2851)
CONTEC Ultrasound Scanner Mobile Trolley Cart Moving Stand, Hand push Use 2017



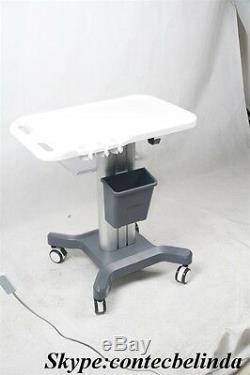



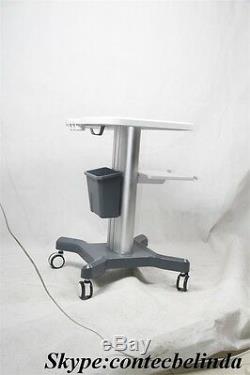


CONTEC Ultrasound Scanner Mobile Trolley Cart Moving Stand, Hand push Use 2017. Thank you for your interesting. This trolley mobile medical cart can be used with Desktop and Laptop B-ultrasound. This link is only for the. Trolley Mobile Cart not including the Ultrasound Scanner and others.
The sale of this item may be subject to regulation by the U. Food and Drug Administration and state and local regulatory agencies.
All of our products are CE approved. If you have questions about legal obligations regarding sales of medical devices, you should consult with the FDA's Center for Devices and Radiological Health. The Fingertip Pulse Oximeter is registered on the Australian Register of Therapeutic Goods (ARTG) with the code 197923, and certified by FDA of United States and CE, TUV of Europe. The Fingertip Pulse Oximeter that is FDA 510K Approved.
(hereinafter referred to as CONTEC) focusing on research, manufacture and distribution of medical instruments, was founded in 1996 as a high-tech company. CONTEC locates in Economic & Technical Development Zone in Qinhuangdao covered an area of 125 acres and building area of over 100000 square meter, which is one of the largest bases for R & D and production of medical devices in China. Contec hopes to cooperate with international companies to supply more innovative design and advanced. We sincerely welcome you to become one of our global partners.
We are looking forward to establishing a successful business relationship with you. The item "CONTEC Ultrasound Scanner Mobile Trolley Cart Moving Stand, Hand push Use 2017" is in sale since Monday, May 22, 2017.
This item is in the category "Business & Industrial\Healthcare, Lab & Dental\Other Healthcare, Lab & Dental". The seller is "madeincontec" and is located in Beijing, China .This item can be shipped worldwide.
- Brand: CON-TEC
- Model: Mobile Trolley Cart Moving Stand
- MPN: 69450401
- Country/Region of Manufacture: China

