
- Home
- Bluetooth
- Brand
- Connectivity
- Memory
- 100 Channels (191)
- 1000 Channels (240)
- 128 Channels (21)
- 200 Channels (6)
- 2500 Channels (77)
- 25000 Channels (25)
- 2gb Ram X 16gb Rom (2)
- 3000 Channels (3)
- 32gb (6)
- 400 Channels (16)
- 500 Channels (20)
- 6000 Channels (8)
- Lots (3)
- Micro Sd 4gb (2)
- Million (5)
- Unknown (6)
- 1800 (7)
- 5500 (4)
- 39000 (2)
- ... (4170)
- Series
- Ds3508 (12)
- Ds3508-er (5)
- Ds3578 (6)
- Ds3608 (10)
- Ds3608-dp (3)
- Ds3608-sr (21)
- Ds4308 (7)
- Ds4308-hd (3)
- Li3608-er (5)
- Memor X3 (6)
- Philips Dpm (5)
- Symbol Ds6707 (8)
- Symbol Ls2208 (3)
- U1-d (4)
- Voyager 1472g (5)
- Voyager Xp 1472g (3)
- Zebra Ds3608 (5)
- Zebra Ds3608-sr (15)
- Zebra Ds9208 (5)
- Zebra Li3608 (18)
- ... (4665)
- Technology
Carejoy Handheld Digital Ultrasound Scanner Machine+Convex&Transvaginal 2 Probes
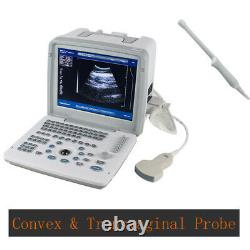
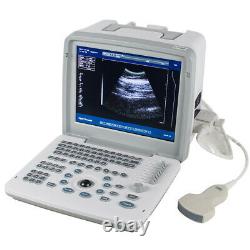
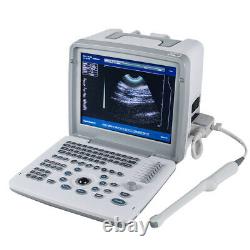





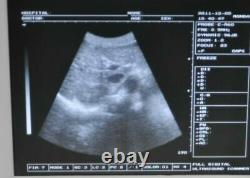


RDA: Real-time dynamic aperture imaging. DRA: Dynamic real-time acoustic apodizer. Ensure that the image is not distorted, edge clearer, level richer. 8 sections TGC gain adjustment. With abundant built-in software packages. Comprehensive software measurement capabilities, to allow more extensive clinical application. Probe connected with a variety of choice to achieve maximum functionality. VLSI, advanced technology, stable performance.
Reasonable cost control, to achieve the perfect embodiment of price and performance. Image magnification: 0.8, 1.0, 1.2, 1.5, 1.8, 2.0. Local zoom: 2 times local zoom in real time. Dynamic range: 64dB - 96dB adjustable.
Focus: 4-segments dynamic electronic focuses selected. Pre-processing: variable aperture, image direction, dynamic filter, edge enhancement, etc. Post-processing: digital space time filter, 8 y corrections, 16 Pseudo Colors, column correlation, frame correlation, spot correlation, linear interpolation, etc.Multi-frequency: 2.5MHz/3.0MHz/3.5MHz/4.0MHz etc. Measurement: distance, circumference/acreage, HR, pregnancy week (BPD, GS, CRL, FL, AC) and calculating fetus weight, etc. Annotation: Chinese/English interface transition; hospital name, doctors/patients name, case number, gender, age; 16 body marks with probe location, full screen character annotation, real time clock display. Puncture lead: Image B: Puncture guide line under B mode. Gain adjustment: 8-segments TGC adjustment, GAIN adjustment or near field, far field, overall gain adjustable.
Image reverse: left/right, black/white, up/down. Storage: 128 images permanent storage. Cine loop: 256 images real time display cycling/one-by-one checked. Output interface: 2 SVGA video outputs, SVGA color monitor circumscribed; 2 PAL video outputs, which can be connected with PAL standard monitor, Video thermal recorder, ultrasound image workstation, etc. 12.1 inch SVGA high resolution monitor.80 elements R60, 3.5MHz multi-frequency convex probe. 3D Ultrasound image working station, etc. User Manual in English:1 each. Statement :The sale of this item may be subject to regulation by the U.
Food and Drug Administration and state and local regulatory agencies. The Fingertip Pulse Oximeter is certified with the US FDA 510K No.
K070371, the CE & TUV of Eureope and it is on the Australian Register of Therapeutic Goods (ARTG) with the code 136606. The Powered Surgical Instrument / Speed 808 System is certified with the US FDA 510(k) Number:K132989. The Powered Surgical Instrument / Hair Remove Device is certified with the US FDA 510(k) Number:K180353. The Powered Surgical Instrument / Hair Remove System is certified with the US FDA 510(k) Number:K141973.Massager, vacuum, light induced heating / Slimming Treatment Device is certified with the US FDA 510(k) Number:K161892. You pay us what you see on the invoice, i. Please make sure it is correct. Or replace item for you free of charge. This item is in the category "Business & Industrial\Healthcare, Lab & Dental\Medical & Lab Equipment, Devices\Ultrasound Machines".
The seller is "superseller66" and is located in this country: CN. This item can be shipped to United States, all countries in Europe, Canada, Australia.
- Brand: Carejoy
- Intended Use/Discipline: Obstetrics & Gynecology
- Mounting: Laptop/Portable
- Model: RUS-9000B
- Country/Region of Manufacture: China
- Warranty: 2 years
- Image mode: B, B/B, 4B, B+M, M
- Image magnification: *0.8, *1.0, *1.2, *1.5, *1.8, *2.0
- Storage: 128 images permanent storage
- Cine loop: 256 images real time display cycling/one-by-one checked
- 1 USB: With
- Connector(s): 2 probe connector
- Output: 2 SVGA video outputs
- Express shipping: 5-7 Days
- DBF: Digital Beam Forming
- RDA: Real-time dynamic aperture imaging
- DRA: Dynamic real-time acoustic apodizer
- Connectors: Two probe connectors
- Display /Monitor / Screen: 12.1-inch SVGA high resolution monitor
- 3D work station software: Yes Free
- Model Theme: Portable Ultrasound Sanner

