
- Home
- Brand
- Features
- Battery Operated (4)
- Channel (3)
- Channel Monitor (42)
- Copier, Portable (8)
- Ctcss (19)
- Dcs (5)
- Digital (7)
- Digital Display (8)
- Digital Handheld (4)
- Gps (9)
- Handy Receiver (3)
- Motorola (4)
- P25 (3)
- Portable (10)
- Portable, Scanner (15)
- Printer (3)
- Radio (13)
- Scanner (17)
- Voice Activated (4)
- Wi-fi (6)
- ... (4631)
- Image Sensor
- Item Width
- Model
- Aor Ar-dv10 (26)
- Bcd325p2 (67)
- Bcd396t (28)
- Bcd396xt (34)
- Bcd436hp (333)
- Bearcat (24)
- Cms600p2 (511)
- Cms600p2-vet (80)
- Cms600p2vet (91)
- Ds3608-sr00003vzcn (23)
- Handscan V8 (26)
- High Resolution (123)
- Pro-106 (39)
- Pro-651 (34)
- Pro-668 (50)
- Pro-96 (32)
- S50a (24)
- Sds100 (203)
- Trx-1 (134)
- Ws1040 (90)
- ... (2846)
- Product Line
CMS600P2/P1 Digital laptop Ultrasound Scanner probes, 5 optional probe, CONTEC




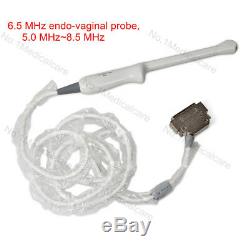




3.5 MHz convex probe, 2.0 MHz5.0 MHz, abdomen. 3.5mhz Micro convex probe, 2.0-5.0mhz, cardiac. 7.5 MHz linear probe, 5.0 MHz10.0 MHz, small organ. 6.5 MHz endo-vaginal probe, 5.0 MHz8.5 MHz 7.5mhz rectal probe, 5.0-10.0mhz, vet for animals. The sale of this item may be subject to regulation by the U.
Food and Drug Administration and state and local regulatory agencies. The item is registered on the Australian Register of Therapeutic Goods(ARTG) with the code 197923, and certified by FDA of United States and CE, TUV of Europe. The Fingertip Pulse Oximeter that is FDA 510K Approved. Make sure you get the item safely.
100% satisfaction is our goal! Client is reaponsible for the custom cleatance and custom duty. Contec Medical Systems focusing on research, manufacture and distribution of medical instruments, was founded in 1992. At present there are more than 1200 employees in our company. Our product line covers a wide range of 13 categories.Most of the domestic hospitals are our customers. Contec hopes to cooperate with international companies to supply more innovative design and advanced technology products. We sincerely welcome you to become one of our global partners. We are looking forward to establishing a successful business relationship with you.
The item "CMS600P2/P1 Digital laptop Ultrasound Scanner probes, 5 optional probe, CONTEC" is in sale since Tuesday, April 10, 2018. This item is in the category "Industrial\Healthcare, Lab & Dental\Medical/Lab Equipment Attachments & Accessories\Medical Sensors & Ultrasound Probes".1medicalcare" and is located in Qinhuang Dao, Hebei Province. This item can be shipped to Australia, North, South, or Latin America, all countries in Europe, all countries in continental Asia, New Zealand.
- Model: CMS600P2 probe
- MPN: 69450401
- Brand: CON-TEC
- Bundle Listing: Yes
- UPC: Does not apply

