
- Home
- Band
- 700 Mhz (2)
- Air (6)
- Am, Fm (6)
- Band (3)
- Cb, Uhf, Vhf (2)
- Digital (2)
- Hf, Uhf, Vhf (2)
- Hf, Vhf, Uhf (18)
- Hf, Vhf, Uhf, Cb (10)
- True I / Q Scanner (3)
- Uhf (169)
- Uhf, Hf (3)
- Uhf, Vhf (22)
- Uhf, Vhf, Cb (2)
- Uhf, Vhf, Hf (197)
- Uhf, Vhf, Hf, Cb (35)
- Uniden (4)
- Vhf (18)
- Vhf, Uhf (6)
- Vhf, Uhf, Cb (9)
- ... (4313)
- Brand
- Isbn
- Model
- Aor Ar-dv10 (26)
- Bcd325p2 (67)
- Bcd396t (28)
- Bcd396xt (34)
- Bcd436hp (334)
- Bearcat (25)
- Cms600p2 (511)
- Cms600p2-vet (80)
- Cms600p2vet (91)
- Ds3608-sr00003vzww (23)
- Handscan V8 (26)
- High Resolution (123)
- Pro-106 (39)
- Pro-651 (34)
- Pro-668 (50)
- Pro-96 (32)
- S50a (24)
- Sds100 (203)
- Trx-1 (134)
- Ws1040 (90)
- ... (2858)
- Type
- Barcode Scanner (173)
- Barcode Scanners (17)
- Barcode Scannner (12)
- Calling System (48)
- Digital Scanner (13)
- Earpiece (9)
- Hand Held (23)
- Handheld (201)
- Handheld Scanner (71)
- Mobile (10)
- Mobile Scanner (10)
- Pager System (11)
- Parts & Accessories (11)
- Portable (20)
- Portable / Handheld (841)
- Radio Scanner (10)
- Radio Scanners (9)
- Scanner (85)
- Ultrasound Scanner (10)
- Walkie-talkie (9)
- ... (3239)
- Weight
Color Doppler Ultrasound Scanner Cardiac Diagnostic Machine Phased Array Probe
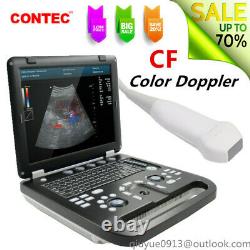

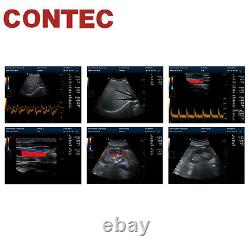








CMS1700A is a color Doppler ultrasonic diagnostic device with high resolution, which has a powerful computer processing platform. The system is mainly suitable for the diagnosis of abdomen, heart, peripheral vessels, breast, obstetrics and gynecology, small organs, urology, muscle, incretion and pediatrics, etc.
It adopts Doppler ultrasound imaging technology, advanced image processing technology such as digital beam-forming technology, tissue harmonic imaging (THI), image speckle suppression, etc. And digital integrated graphic management system, and the internal professional measurement software package can fully meet the clinic diagnostic requirement.
1Color Doppler (CF), color power Doppler imaging (PDI). 2Extended interface: VIDEO interface, S-VIDEO interface, RJ-45 interface, USB interface, VGA interface. Type of protection against electric shock: class equipment. Degree of protection against electric shock: type B applied part. Operating voltage: AC 100 V240 V.
Operating frequency: 50 Hz/60 Hz. One abdominal probe (3.5 MHz). Dimension: 370 mm (L) × 360 mm (W) × 80 mm (H). Food and Drug Administration and state and local regulatory agencies. If you have questions about legal obligations regarding sales of medical devices, you should consult with the FDA's Center for Devices and Radiological Health. The Fingertip Pulse Oximeter is registered on the Australian Register of Therapeutic Goods (ARTG) with the code 197923, and certified by FDA of United States and CE, TUV of Europe. The Fingertip Pulse Oximeter that is FDA 510K Approved. The item "Color Doppler Ultrasound Scanner Cardiac Diagnostic Machine Phased Array Probe" is in sale since Tuesday, March 30, 2021. This item is in the category "Business & Industrial\Healthcare, Lab & Dental\Medical & Lab Equipment, Devices\Ultrasound Machines". The seller is "qiuyu_4054" and is located in Qinhuangdao. This item can be shipped worldwide.- Mounting: Laptop/Portable
- MPN: 69450401
- Brand: CONTEC
- Intended Use/Discipline: Biological Laboratory
- Ultrasound Machine Features: Color Doppler

