
- Home
- Brand
- Connectivity
- Features
- Battery Operated (4)
- Channel (3)
- Channel Monitor (42)
- Copier, Portable (8)
- Ctcss (19)
- Dcs (5)
- Digital (7)
- Digital Display (8)
- Digital Handheld (4)
- Gps (9)
- Handy Receiver (3)
- Motorola (4)
- P25 (3)
- Portable (9)
- Portable, Scanner (15)
- Printer (3)
- Radio (13)
- Scanner (17)
- Voice Activated (4)
- Wi-fi (6)
- ... (4624)
- Memory
- Model
- Aor Ar-dv10 (26)
- Bcd325p2 (67)
- Bcd396t (28)
- Bcd396xt (34)
- Bcd436hp (333)
- Bearcat (24)
- Cms600p2 (511)
- Cms600p2 Vet (23)
- Cms600p2-vet (80)
- Cms600p2vet (91)
- Handscan V8 (26)
- High Resolution (123)
- Pro-106 (39)
- Pro-651 (34)
- Pro-668 (50)
- Pro-96 (32)
- S50a (24)
- Sds100 (203)
- Trx-1 (134)
- Ws1040 (90)
- ... (2838)
- Type
- Barcode Scanner (173)
- Barcode Scanners (17)
- Barcode Scannner (12)
- Calling System (48)
- Digital Scanner (13)
- Earpiece (9)
- Hand Held (23)
- Handheld (201)
- Handheld Scanner (69)
- Mobile (10)
- Mobile Scanner (10)
- Pager System (11)
- Parts & Accessories (11)
- Portable (20)
- Portable / Handheld (838)
- Radio Scanner (10)
- Radio Scanners (9)
- Scanner (84)
- Ultrasound Scanner (10)
- Walkie-talkie (9)
- ... (3223)
Handheld Ultrasound Scanner Digital Machine w Linear Probe Free Box & Battery
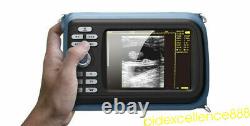

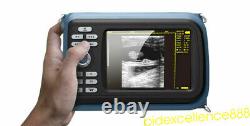





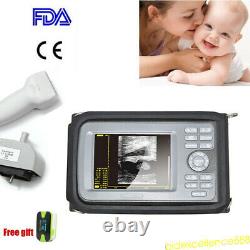


Hand type Handheld Ultrasound Scanner Digital Machine+ Micro-convex 5.5 Human. Display Modes: B, B+B, B+M, M, 4B. Image Conversion: distance, circumference, area, volume, EF rate, heart.
Measurements: BPD, GS, CRL, FL, HC, AC, EDD, GA, FWVIDEO (PLAD, NTSC), USB2.0. One(1) L40/7.5Mhz HF linear Probe.
Comments: date&time, name, sex, age, hospital. Applications:Professional sofeware package in Obstetrics, Gynecology, Urology and Small parts. Statement:The sale of this item may be subject to regulation by the U. Food and Drug Administration and state and local regulatory agencies. The Fingertip Pulse Oximeter is certified with the US FDA 510K No.
K070371, the CE & TUV of Europe and it is on the Australian Register of Therapeutic Goods (ARTG) with the code 136606. The Powered Surgical Instrument / Speed 808 System is certified with the US FDA 510(k) Number:K132989. The Powered Surgical Instrument / Hair Remove Device is certified with the US FDA 510(k) Number:K180353.
The Powered Surgical Instrument / Hair Remove System is certified with the US FDA 510(k) Number:K141973. Massager, vacuum, light induced heating / Slimming Treatment Device is certified with the US FDA 510(k) Number:K161892. Or replace item for you. W e commit ourself to do 100% customer satisfactione. We'll do our best to help you. This item is in the category "Business & Industrial\Healthcare, Lab & Dental\Medical & Lab Equipment, Devices\Ultrasound Machines". The seller is "bidexcellence888" and is located in this country: CN. This item can be shipped to United States, Canada, United Kingdom, Germany, France, Australia.
- Modified Item: No
- Country/Region of Manufacture: China
- Custom Bundle: No
- Image Color: Black & White
- Brand: Carejoy
- Intended Use/Discipline: Obstetrics & Gynecology
- UPC: 190891515230

